|
1X HBS-P+ Buffer (GE Healthcare) was used for CD64, CD32a, and CD16a binding assays. Multi-concentration binding kinetic experiments were performed on an Octet RED96 instrument at 30☌. On the basis of the nonreduced ADC chromatogram, percent areas of DAR 0, 1, and 2 species were quantified using Agilent ChemStation Software, and DAR was calculated using the following equation: DAR = (% area of DAR1 + % area of DAR2 × 2)/100. Nonreduced and reduced samples were injected at 7 μg per load. For reduced samples, mAb and ADC were treated with 20 mmol/L dithiothreitol in 6 mol/L guanidine-HCl and 50 mmol/L TRIS-HCl, pH 7.8 at 56☌ for 30 minutes prior to high-performance liquid chromatography (HPLC) analysis. The column was kept at 75☌ and flow rate was maintained at 1 mL/minute. The column was first equilibrated at 70% mobile phase A (0.1% TFA in water) and 30% mobile phase B (0.1% TFA in acetonitrile) for 1 minute, followed by a linear gradient from 30% B to 50% B over 20 minutes where the analyte eluted. MAb and ADC samples were analyzed on a ZORBAX StableBond C3 Column (Agilent Technologies, catalog no.: 863973-909) using an Agilent 1100 HPLC System with UV detection at 214 nm. The encouraging preclinical data support the further development of ARX788 for treatment of patients with HER2-positive breast and gastric cancer, including those who have developed T-DM1 resistance, and patients with HER2-low expression tumors who are currently ineligible to receive HER2-targeted therapy. Breast and gastric cancer patient-derived xenograft studies confirmed strong antitumor activity of ARX788 in HER2-positive and HER2-low expression tumors, as well as in a T-DM1-resistant model. 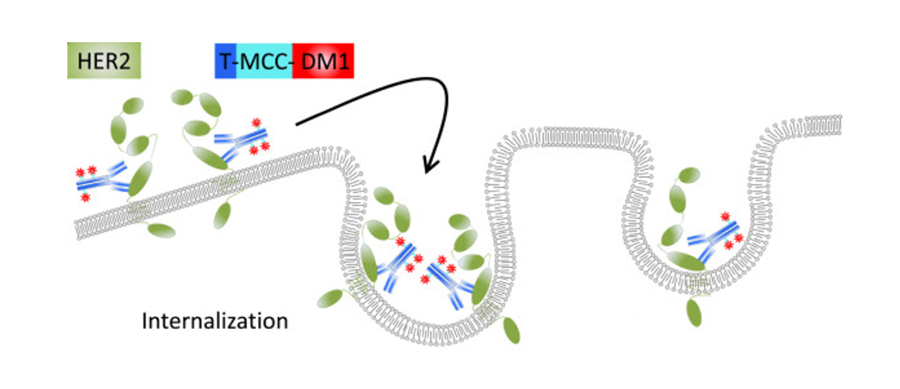
Similarly, ARX788 significantly inhibited tumor growth, and generally outperformed T-DM1 in HER2-high and HER2-low expression xenograft models. When compared in vitro against T-DM1 across a panel of cancer cell lines, ARX788 showed superior activity in the lower HER2-expressing cell lines and no activity in normal cardiomyocyte cells. ARX788 exhibits high serum stability in mice and a relatively long ADC half-life of 12.5 days. ARX788 is a next-generation, site-specific anti-HER2 ADC that utilizes a unique nonnatural amino acid–enabled conjugation technology and a noncleavable Amberstatin (AS269) drug-linker to generate a homogeneous ADC with a drug-to-antibody ratio of 1.9. First-generation antibody–drug conjugates (ADC) are heterogeneous mixtures that have shown clinical benefit, but generally exhibited safety issues and a narrow therapeutic window due, in part, to off-target toxicity caused by ADC instability.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
